Hydrogen: Artificial Photosynthesis & a Clean Fuel
6 minute read
Updated on: 29 Aug 2021
In the last chapter, we saw that batteries allow us to replace 71% of oil-based fuels with electricity. This is for cars, trucks, and other land transport, but batteries can’t hold enough energy per kg to make airplanes or ships electric
.
Hydrogen can help here and, once cheap enough, it could also be used to fuel cars! How? Hydrogen holds 128x as much energy as Li-Ion batteries per kilogram
.
In nature, hydrogen (H) usually appears as a pair of two atoms (H₂). This is also true for oxygen (O₂).
Water (H₂O) is made of hydrogen and oxygen. You’ve probably never seen water fall apart into H₂ and O₂, right? That’s because water is a stable compound. Splitting H₂O requires energy. Therefore, we can store energy by splitting water (2H₂O) into 2H₂ and O₂
and later get the energy back by bringing them back together.
Fuel from water?
The idea is simple: use clean electricity (from renewables or nuclear) to split water in a process called electrolysis.
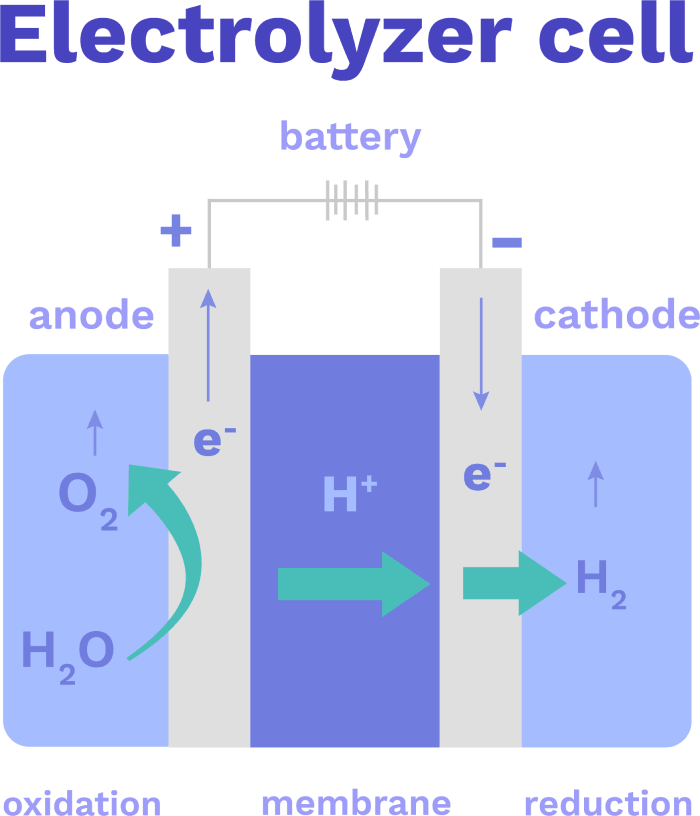
Electrolysis
Later, we can turn the H₂ back into electricity by using what’s called a hydrogen fuel cell:
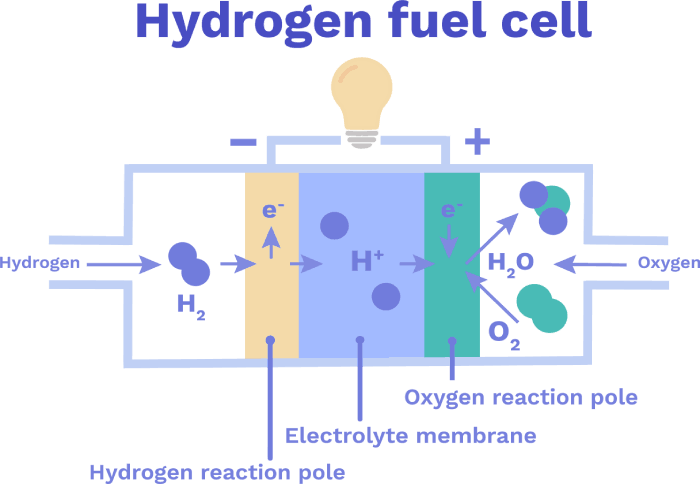
Hydrogen fuel cell
How is hydrogen produced today?
There has been significant demand for hydrogen since 1975.And because hydrogen today is made from natural gas, producing it causes 2% of global CO₂ emissions
!
Why use gas over renewables? Cost. But with innovation and further deployment of solar and wind, we could get costs down by a lot:
What needs to change?
Higher electrolysis efficiency: The more hydrogen per unit of energy, the better!
Higher compression efficiency: Every unit of energy we spend on compression essentially goes to waste, but we need to compress hydrogen to fit it into tanks. It’s a gas, in the end.
Higher fuel cell efficiency: The more energy we get out of each kilogram of hydrogen, the better.
Cheaper electrolysis: Energy is only a part of the cost of hydrogen. Electrolyzers need to become 20-50% cheaper to fulfill the prediction from the graph we showed before
.
Infrastructure: Gases are hard to transport. To avoid the huge upfront cost of building pipelines, H₂ could be produced near to where it’s used. This is a trade off because building many smaller electrolyzers would likely be more expensive than a few big ones
.
Can Artificial photosynthesis help?
What if we could do what plants do to make energy, but do it better?





